About TEDD
Tissue Engineering for Drug Development and Substance Testing
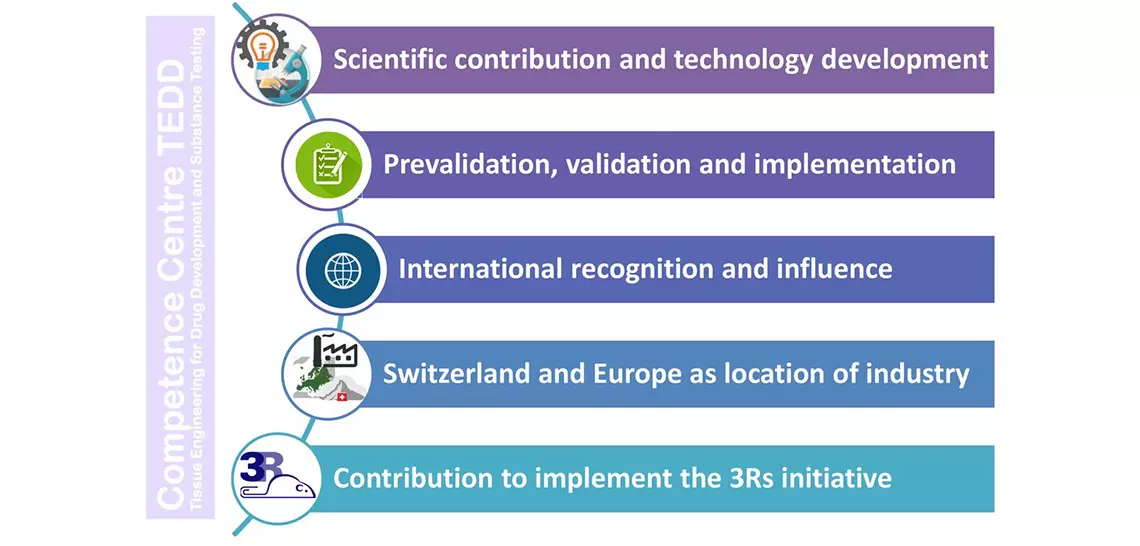
Goals
Collaborative platform
TEDD was established together with the introduction of 3D cell cultures technologies to the wider audience. The goal of us is to support the development of these complex tissue systems and implement it into the routine application. In order to maximize the success, a cooperation of partners with complementary skills –academia, industry (in particular biotechnology, pharmaceutical, medtech and cosmetics companies) and clinics is required. 3D cell culture became a widely acceptable and desirable technology, however, it did not yet reach its full potential. Therefore, further efforts should be made to maintain the progress of 3D cell technologies development. The goal of us is to support the 3R principles implementation and the introduction of 3D cell culture technologies as the alternative for animal testing which can be used in your company.
3D organotypic cell culture technologies

Cell cultures are an invaluable technology used widely from basic research to industrial applications. The conventional 2D cell culture systems, based on cell lines and primary cells, has been used for a long time to understand the complex cellular physiology i.e. on how cells function and respond to stimuli. They have been also used in regenerative medicine, production of substances such as antibodies and in the pharmaceutical and cosmetic industry to test compounds and drug candidates. However, technological advancements are necessary to address emerging complex challenges of the traditional systems.
Physiological relevance is a key parameter for improving the predictive power of cell-based technologies. To achieve in vivo like features in in vitro cell culture, recently three dimensional (3D) culture systems were introduced. 3D cell cultures recapitulate tissue architecture, cell-cell and cell-matrix interactions in a physiological manner. This area is one of the fastest growing experimental approaches in life sciences. The three-dimensional cell culture market was $438.1 million in 2013. The market was expected to grow to $586.1 million in 2014 and about $2.2 billion in 2019, with a compound annual growth rate (CAGR) of 30.1% (3D Cell Culture: Technologies and Global Markets. (Jan 2015) BCC Research Biotechnology Report).
3D for 3Rs

Animals are currently used in studies, where an absolute requirement for tissue-specific information is needed, such as in drug efficacy and toxicity testing. For example in cancer research, the tumour is established in rodent models by either surgically implanting tumour cells, patient-derived xenografts (PDX) or creating genetically-engineered animals that spontaneously develop human-like tumours. However, animal testing often fails to predict the effects of drugs on a human. Moreover, the use of animals in research, teaching and testing is an important ethical and political issue, because many of these experiments cause pain to the animals involved or reduce their quality of life.
These facts are motivating governments and regulatory organizations around the globe to support the implementation of alternative methods for animal testing. 3R principles (Replacement, Reduction, and Refinement) have subsequently become embedded in national and international legislation regulating the use of animals in scientific procedures. According to the Animal Protection Index Switzerland, together with the UK, New Zealand and Austria, has the strictest animal protection laws in the world, including the use of animals in scientific research. In 2018 compared with 2009, the number of animals used in studies has fallen by more than 16.9%. The Animal Protection Act requires assessment and approval of individual experiments involving animals. However, according to the official statement, animal experiments remain essential for developing new drug therapies and cannot be replaced in all cases by alternative methods.
3D organotypic cell culture technologies have the potential to replace animal testing and provide more reliable data as they bridge the gap between cell and human tissues. 3D cultures may greatly improve cell-based drug screening and identify toxic and ineffective substances at an earlier stage of the drug discovery pipeline than animal or clinical trials.
At TEDD we see a crucial time in Switzerland now to make a change, as the public’s opinion is more and more engaged and aware of ethical concerns of animal testing. Furthermore, in 2020 the Swiss public will vote for the total ban of animal experiments in research and drug development. Thus, we believe that TEDD has a pivotal role in disseminating the information on alternative methods. At the same time the government and regulatory agencies need to introduce structured efforts to make this happen: by providing financial resources and developing guidelines for the validation and harmonisation of organotypic models. TEDD sees its role in bridging academia with the biomedical sector since biotech and pharma have a powerful lobby, and they dramatically influence the political decision in the country.
Competencies
We combines diverse skills through integrative cooperation among the partners to form a powerful and successful network.
Our competencies include:
- 3D cell and tissue models
- Assay development
- Imaging technologies
- Automation
- Molecular reporter systems
- Standardization and quality standards
- Biomimetic scaffold substances
- Bioprinting
TEDD Activities
The competence center is an important platform from which 3D cell culture technology can be actively influenced. Regular events and activities at national and international level ensure exchange and progress.
TEDD Vision
Structure and history
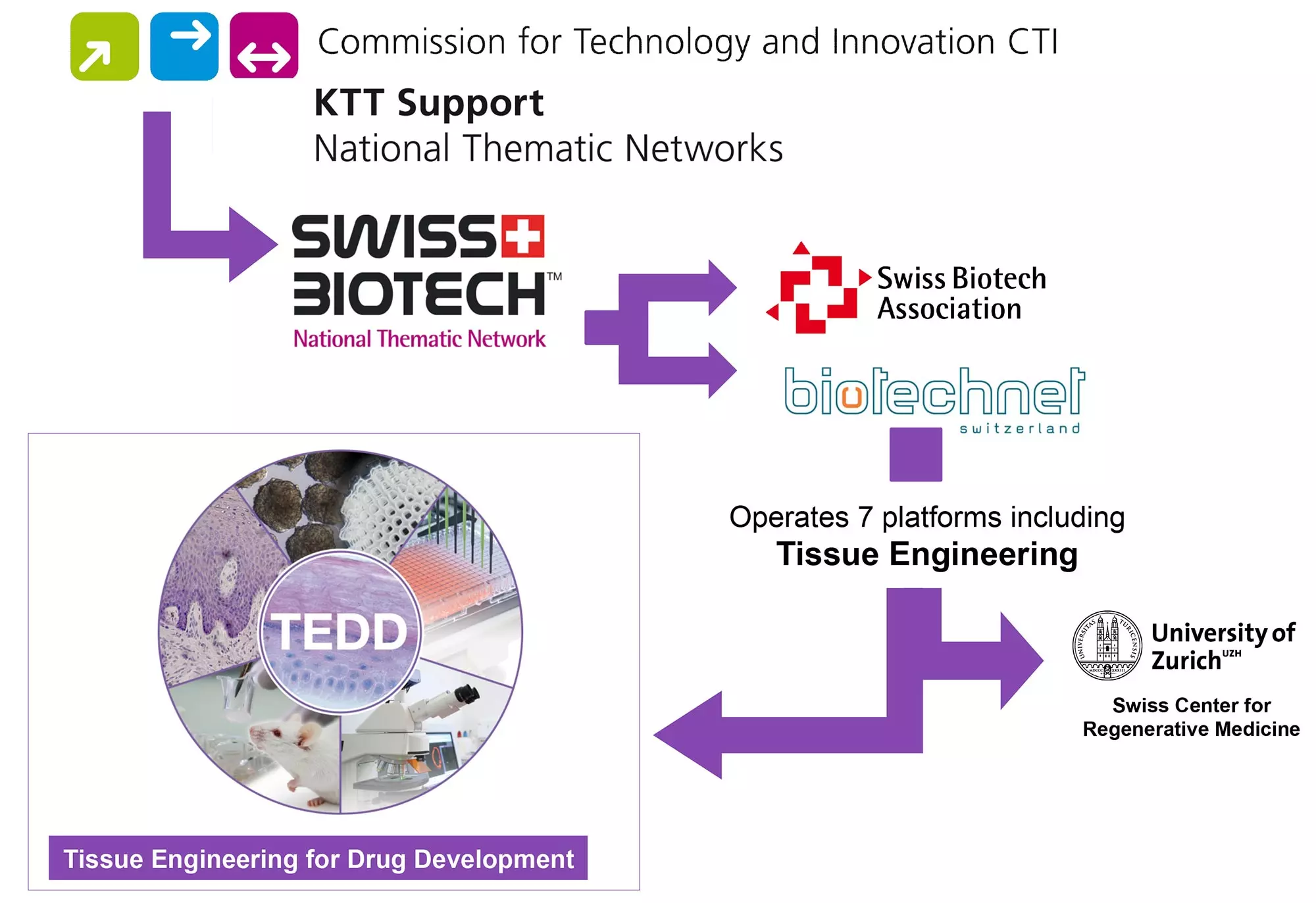
The national Competence Centre Tissue Engineering for Drug Development and Substance Testing TEDD was founded in 2011 by the members of section «Tissue Engineering» at the Zurich University of Applied Sciences (ZHAW) lead by Prof. Ursula Graf-Hausner and company InSphero AG, Swiss leading supplier of organotypic in vitro 3D microtissues for drug testing.
TEDD received initial funding from Gebert Ruef Foundation . In 2010 the project was one of the winners of the annual call for projects «BREF – Brückenschläge mit Erfolg» (building bridges successfully). TEDD is embedded in the ZHAW, in the national consortium for biotechnology biotechnet Switzerland and the National Thematic Network (NTN) Swiss Biotech.
Partner networks

TEDD Competence Centre as member belongs to: