Section for Organic and Medicinal Chemistry

«With our medicinal chemistry expertise, structure-based design and organic synthesis, we develop new treatment options for patients.»
Head of Section for Organic and Medicinal Chemistry
The Organic and Medicinal Chemistry group has many years of experience in both academic and industrial drug discovery. We design and synthesize new small molecule and peptide drug molecules for diseases that are currently untreatable. In this way, we lay the foundation for innovations in pharmaceutical research and development. In the Competence Center Drug Discovery, we maintain an interdisciplinary network and in recent years have contributed to innovations in the fight against brain tumors, resistant pathogenic bacteria, leukemia, skin cancer and SARS-CoV-2, among others.
Competencies & Research
Medicinal Chemistry

Our Medicinal Chemistry activities span from hit identification, through hit to lead development and lead optimization to delivering active pharmaceutical ingredients (APIs) for in vitro and in vivo studies. Our focus is on the development of clinical candidates with our industry partners and includes the structural analysis of the target proteins (and their complexes) and results in the delivery of novel and patentable small organic ligands for clinical trials by using a combination of classical SAR profiling and state of the art fragment-based approaches.
- structural analysis of the target protein (X-ray analysis of co-crystal structures)
- NMR binding studies (ligand/protein)
- fragment-based design of novel ligands
- effective application of industry standard docking software
- multi-step organic synthesis of ligands
- establishment of structure activity relationships (SAR)
- development of data bank based IT solutions for the information management of chemical and biological data (SAR, inventory, etc.)
Beyond we offer a multitude of possible R & D collaborations:
- collaboration with research based pharmaceutical companies according to our complete set of medicinal chemistry capacities (from structure based design to organic synthesis and the delivery of small molecule clinical drug candidates)
- consulting in the area of medicinal chemistry and process management for start-up as well as small and mid-sized pharmaceutical companies (computational drug design, establishing screening concepts, development of structure activity relationships, development of clinical candidates, process optimization employing SixSigma process management tools)
Organic Synthesis + Synthetic Methodology

Our activities in the area of Organic Synthesis and Synthetic Methodology are focused around the multi-step syntheses of catalysts and APIs. Our core competence in Organic Synthesis and implementation of computational chemistry allow for a flexible research strategy for catalyst development and drug discovery. This flexible and multi-faceted approach has been applied with great success to the benefit of our industry
partners.
In addition to classical organic synthetic techniques we have invested in advanced organic synthetic technologies such as microwave reactors, microreactors, parallel synthesis abd automated solid phase synthesizers.
Organic Analysis & Structure Elucidation

We have the expertise and equipment for Analyses and Structure Elucidation of Organic Molecules of both routine in house samples or those of our customers, as well as ongoing research activities in the isolation, identification and optimization of biogenic drug molecules.
Cheminformatics
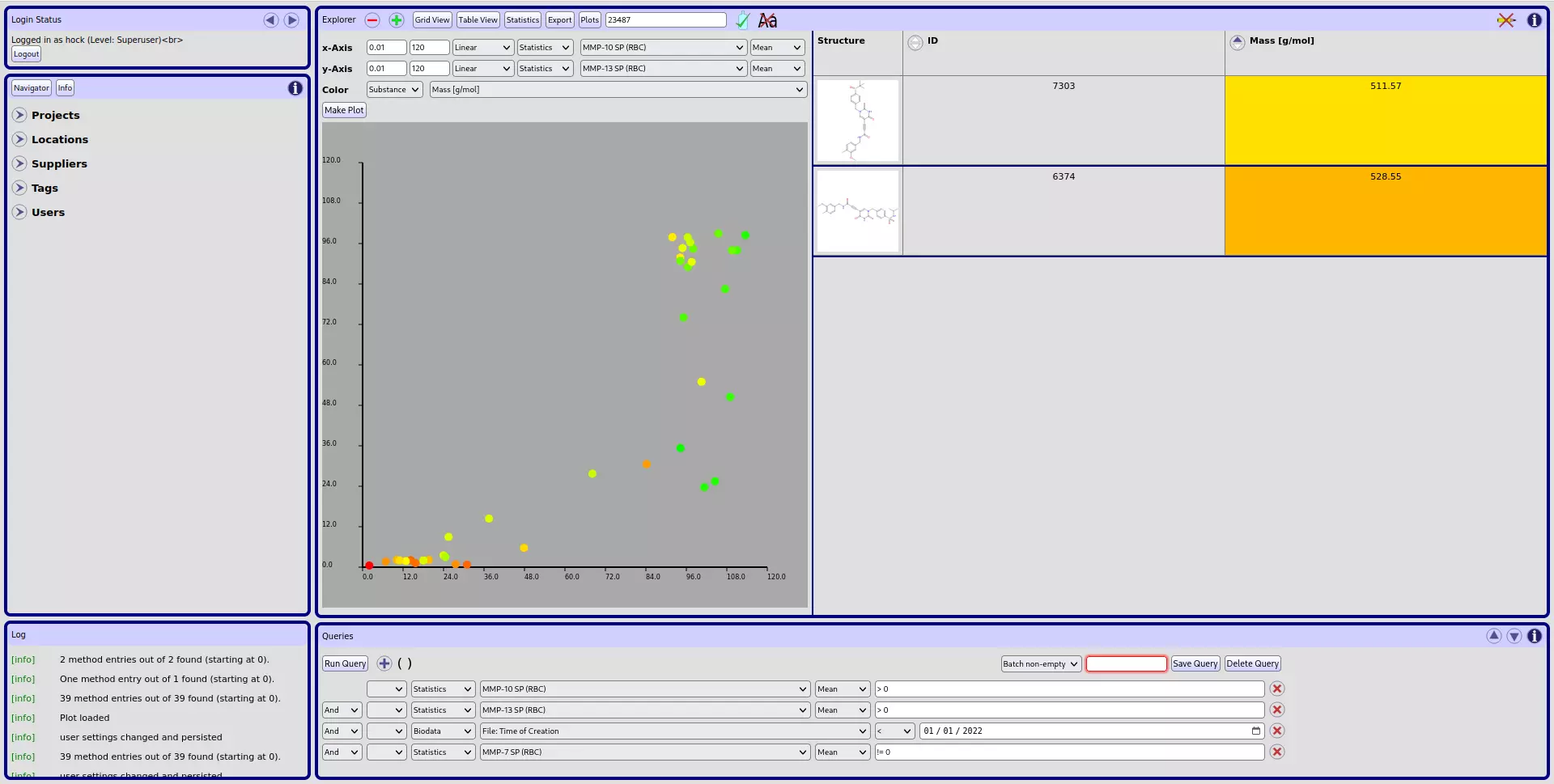
The goal of our activities in Cheminformatics is the development of IT solutions for the storage and convenient analysis of large data sets. Those data sets usually arise from medicinal chemistry projects. Here, the analysis of chemical structures in combination with effective and customizable displays of biological data are particularly useful, but large chemical inventory data sets of chemical organizations are also easily handled.
CyBy2: We offer you a adaptation of our CyBy2 IT-solution (a structure-based data management tool) as the central tool for your needs regarding data storage and data analysis of chemical and biological data sets.
Technologies & Equipment

- Thermo MSQ Plus UHPLC‐single quadrupole system
- Liberty Blue Automated Microwave Peptide Synthesizer
- Bruker AVANCE III HD 500MHz OneBay NMR Spectrometer
- Waters Xevo-Acquity UPLC-triple quadrupole system
- Gilson PLC 2020 Purification system
- LC-MS Agilent Ion Trap XCT

- lab reactor LabMax: automatic synthesis (-40 °C to 140 °C)
- ChemSpeed PSW 1100: automatic peptide synthesis equipment
- Biotage, MLS System: microwave assisted synthesis equipment
- Büchi autoclave systems: up to 50 bar and maximum 1 litre
- industry sized evaporation system Büchi R-220
- Perkin-Elmer 341 polarimeter, FTIR, GC

- Knauer preparative HPLC System: flow rate up to 500 ml/min
- Ehrfeld and Little Things Factory semi-automatic microreactor synthesis equipment
- Radleys parallel synthesis equipment
- ISCO chromatography system
- GeneVac EZ-2 evaporation system
- 6-Core workstation equipped with molecular docking software
Projects
-
BRIDGE Small Molecules against Leukemia
The aim of this project is to develop small molecules for the treatment of acute myeloid leukemia.
-
Evaluation of the potential of PROTACs
The goal of this project is to evaluate within a SNF Spark project the potential of Protacs.
-
Evaluation of the potential of targeting the IFN1 pathway
The goal of this project is to evaluate within a SNF Spark project the potential of targeting the IFN1 pathway.
Selected publications
- Antiprotozoal Structure–Activity Relationships of Synthetic Leucinostatin Derivatives and Elucidation of their Mode of Action / M. Brand, L. Wang, S. Agnello, S. Gazzola, F. M. Gall, L. Raguž, M. Kaiser, R. S. Schmidt, A. Ritschl, J. Jelk, A. Hemphill, P. Mäser, P. Bütikofer, M. Adams, R. Riedl, Angew. Chem. Int. Ed. 2021, 60, 15613.
- Drug Design Inspired by Nature: Crystallographic Detection of an Auto‐Tailored Protease Inhibitor Template / F. M. Gall, D. Hohl, D. Frasson, T. Wermelinger, P. R. E. Mittl, M. Sievers, R. Riedl, Angew. Chem. Int. Ed. 2019, 58, 4051.
- A Structural View on Medicinal Chemistry Strategies against Drug Resistance / S. Agnello, M. Brand, M. F. Chellat, S. Gazzola, R. Riedl, Angew. Chem. Int. Ed. 2019, 58, 3300.
- Pseudouridimycin: The First Nucleoside Analogue That Selectively Inhibits Bacterial RNA Polymerase / M. F. Chellat, R. Riedl, Angew. Chem. Int. Ed. 2017, 56, 13184.
- Targeting Antibiotic Resistance / Chellat, Mathieu; Raguž, Luka; Riedl, Rainer - Angew. Chem. Int. Ed. 2016, 55, 6600-6626; Angew.Chem. 2016, 128, 6710–6738.
- Molecular recognition of the catalytic zinc (II) ion in MMP-13: Structure-based evolution of an allosteric inhibitor to dual binding mode inhibitors with improved lipophilic ligand efficiencies / Fischer, Thomas; Riedl, Rainer - invited article for the Special Issue "Enzyme-Inhibitor Interaction as Examples of Molecular Recognition" Int. J. Mol. Sci. 2016, 17, 314. Front cover story 3/2016.
- Merging Allosteric and Active Site Binding Motifs: De novo Generation of Target Selectivity and Potency via Natural-Product-Derived Fragments / Lanz, Jan; Riedl, Rainer - ChemMedChem. 2015, 10, 451–454. Front cover story 3/2015.
Publications
-
Sabani, Besmira; Brand, Michael; Albert, Ina; Inderbitzin, Joelle; Eichenseher, Fritz; Schmelcher, Mathias; Rohrer, Jack; Riedl, Rainer; Lehmann, Steffi,
2023.
Nanomedicine: Nanotechnology, Biology and Medicine.
47(102607).
Available from: https://doi.org/10.1016/j.nano.2022.102607
-
Bagatella, Stefano; Haghayegh Jahromi, Neda; Monney, Camille; Polidori, Margherita; Gall, Flavio Max; Marchionatti, Emma; Serra, Fabienne; Riedl, Rainer; Engelhardt, Britta; Oevermann, Anna,
2022.
Journal of Neuroinflammation.
19(304).
Available from: https://doi.org/10.1186/s12974-022-02653-1
-
Kiss, Cedric; Gall, Flavio M.; Dreier, Birgit; Adams, Michael; Riedl, Rainer; Plückthun, Andreas; Mittl, Peer R. E.,
2022.
Structure of a hydrophobic leucinostatin derivative determined by host lattice display.
Acta Crystallographica. Section D: Structural Biology.
78(12), pp. 1439-1450.
Available from: https://doi.org/10.1107/S2059798322010762
Awards & News
ZHAW News-Release 2018: Bachelor thesis on structural optimization of a matrix metalloproteinase-13 inhibitor is awarded Dr. Max Lüthi Prize of the SCS.
ZHAW News-Release 2016: Bachelor thesis on rational drug design and synthesis of cyclic peptides is awarded Dr. Max Lüthi Prize of the SCS.
Poster(PDF 891,1 KB) 2013: SCG-FH Award Category «Molecules for the Life Sciences» Tackling antibiotic resistance: A joint project of the center for organic and medicinal chemistry ZHAW and the Bioversys AG.
ZHAW press release(PDF 74,7 KB) (German) 2013: SCS awarded the Dr. Max Lüthi award for bachelor thesis on the rational design and synthesis of novel protease inhibitors.
ZHAW press release(PDF 46,8 KB) (German) 2012: MSc thesis on the structure based design and synthesis of novel protease inhibitors received Bodenseeinnovation award.
ZHAW press release (PDF 46,4 KB)(German) 2011: Bachelor thesis on development of active substances to combat antibiotic resistance is awarded Dr. Max Lüthi award of the SCS.