Section for Organic and Medicinal Chemistry

«With our medicinal chemistry expertise, structure-based design and organic synthesis, we develop new treatment options for patients.»
Prof. Dr. Rainer Riedl
Head of Section for Organic and Medicinal Chemistry
The Organic and Medicinal Chemistry group has many years of experience in both academic and industrial drug discovery. We design and synthesize new small molecule and peptide drug molecules for diseases that are currently untreatable. In this way, we lay the foundation for innovations in pharmaceutical research and development. In the Competence Center Drug Discovery, we maintain an interdisciplinary network and in recent years have contributed to innovations in the fight against brain tumors, resistant pathogenic bacteria, leukemia, skin cancer and SARS-CoV-2, among others.
Competencies & Research
Medicinal Chemistry

Our Medicinal Chemistry activities span from hit identification, through hit to lead development and lead optimization to delivering active pharmaceutical ingredients (APIs) for in vitro and in vivo studies. Our focus is on the development of clinical candidates with our industry partners and includes the structural analysis of the target proteins (and their complexes) and results in the delivery of novel and patentable small organic ligands for clinical trials by using a combination of classical SAR profiling and state of the art fragment-based approaches.
- structural analysis of the target protein (X-ray analysis of co-crystal structures)
- NMR binding studies (ligand/protein)
- fragment-based design of novel ligands
- effective application of industry standard docking software
- multi-step organic synthesis of ligands
- establishment of structure activity relationships (SAR)
- development of data bank based IT solutions for the information management of chemical and biological data (SAR, inventory, etc.)
Beyond we offer a multitude of possible R&D collaborations:
- collaboration with research based pharmaceutical companies according to our complete set of medicinal chemistry capacities (from structure based design to organic synthesis and the delivery of small molecule clinical drug candidates)
- consulting in the area of medicinal chemistry and process management for start-up as well as small and mid-sized pharmaceutical companies (computational drug design, establishing screening concepts, development of structure activity relationships, development of clinical candidates, process optimization employing SixSigma process management tools)
Organic Synthesis + Synthetic Methodology

Our activities in the area of Organic Synthesis and Synthetic Methodology are focused around the multi-step syntheses of catalysts and APIs. Our core competence in Organic Synthesis and implementation of computational chemistry allow for a flexible research strategy for catalyst development and drug discovery. This flexible and multi-faceted approach has been applied with great success to the benefit of our industry
partners.
In addition to classical organic synthetic techniques we have invested in advanced organic synthetic technologies such as microwave reactors, microreactors, parallel synthesis and automated solid phase synthesizers.
Organic Analysis & Structure Elucidation

We have the expertise and equipment for Analyses and Structure Elucidation of Organic Molecules of both routine in house samples or those of our customers, as well as ongoing research activities in the isolation, identification and optimization of biogenic drug molecules.
Cheminformatics
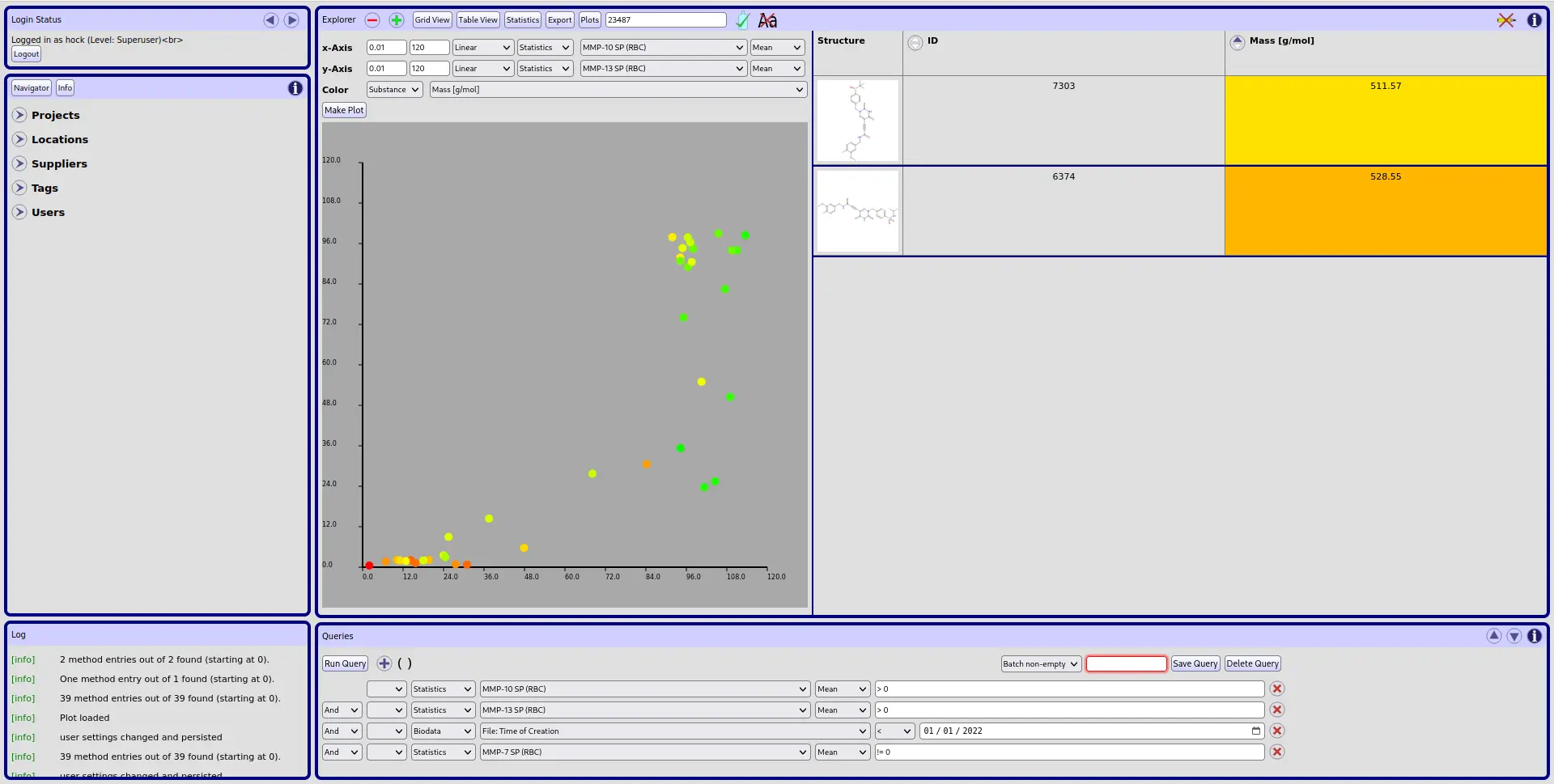
The goal of our activities in Cheminformatics is the development of IT solutions for the storage and convenient analysis of large data sets. Those data sets usually arise from medicinal chemistry projects. Here, the analysis of chemical structures in combination with effective and customizable displays of biological data are particularly useful, but large chemical inventory data sets of chemical organizations are also easily handled.
CyBy2: We offer you a adaptation of our CyBy2 IT-solution (a structure-based data management tool) as the central tool for your needs regarding data storage and data analysis of chemical and biological data sets.
Technologies & Equipment

- Thermo MSQ Plus UHPLC‐single quadrupole system
- Liberty Blue Automated Microwave Peptide Synthesizer
- Bruker AVANCE III HD 500MHz OneBay NMR Spectrometer
- Waters Xevo-Acquity UPLC-triple quadrupole system
- Gilson PLC 2020 Purification system
- LC-MS Agilent Ion Trap XCT

- lab reactor LabMax: automatic synthesis (-40 °C to 140 °C)
- ChemSpeed PSW 1100: automatic peptide synthesis equipment
- Biotage, MLS System: microwave assisted synthesis equipment
- Büchi autoclave systems: up to 50 bar and maximum 1 litre
- industry sized evaporation system Büchi R-220
- Perkin-Elmer 341 polarimeter, FTIR, GC

- Knauer preparative HPLC System: flow rate up to 500 ml/min
- Ehrfeld and Little Things Factory semi-automatic microreactor synthesis equipment
- Radleys parallel synthesis equipment
- ISCO chromatography system
- GeneVac EZ-2 evaporation system
- 6-Core workstation equipped with molecular docking software
Projects
-
Small molecules targeting CD93-signaling for the treatment of acute myeloid leukemia
The aim of this project is to develop small molecules for the treatment of acute myeloid leukemia.
current, 04/2024 - 03/2028
-
Evaluation of the potential of PROTACs
The goal of this project is to evaluate within a SNF Spark project the potential of Protacs.
expired, 04/2024 - 10/2025
-
Peptidic active ingredients II
The aim of this project is to synthesize peptidic active ingredients.
expired, 02/2024 - 11/2025
-
Targeting Gram-negative bacteria
In this project, novel small molecule antibiotics will be developed to target highly resistant Gram-negative bacteria. Gram-negative bacteria can cause various infections such as dermatitis, blood poisoning or pneumonia.
expired, 10/2023 - 08/2025
-
Innovative Immunotherapy Approach
The goal of this project is the synthesis of peptide loaded nanoparticles.
expired, 09/2023 - 02/2024
-
Therapy for the NRAS melanoma subtype
Melanoma is the fifth most common cancer in Switzerland and the most lethal skin cancer. NRAS (neuroblastoma ras viral oncogene homolog)-mutated melanoma is the most aggressive subtype, with no effective therapy after immunotherapy failure. The goal of this project is to develop the first effective…
expired, 02/2023 - 07/2024
-
Platform for drug target identification
A major problem in medicinal chemistry and drug discovery projects is the unknown identity of the molecular biological target, even when very potent ligands are available, such as those discovered in a phenotypic screen. Therefore, methods for hit optimization, such as structure-based design, or…
expired, 01/2023 - 12/2023
-
Inhibitors targeting SARS-CoV-2 proteases
So far, vaccines are available to combat the Covid-19 pandemic, which can protect against a severe course, although part of society also does not want to be vaccinated or cannot do so due to pre-existing conditions. With a drug that can fight the virus in the organism, infected patients with severe…
expired, 10/2022 - 03/2024
-
Developing novel and effective therapies for treatment of childhood brain cancers
Brain cancers are a leading cause of mortality in children. Brainstem glioma constitutes 10% of all pediatric central nervous system tumors, the most common type being a high grade diffuse midline glioma (DMG). Preclinical discoveries fail to translate into clinical success due to: i) drug systemic…
expired, 03/2022 - 08/2023
-
Development of new therapeutic approaches for the treatment of cancer
Further therapeutic approaches are urgently needed for the treatment of cancer. The aim of this project is to synthesize active substances that interact with biological target proteins relevant in cancer therapy. For this purpose, multi-step organic syntheses are carried out and the active…
expired, 11/2021 - 05/2023
-
Developing a gasotransmitter based treatment for pathological skin and tendon calcification
expired, 10/2021 - 09/2023
-
Plants and Minerals in Byzantine Popular Pharmacy. A New Multidisciplinary Approach
Historical texts containing information of the medicinal use of plants, minerals and other natural products are highly interesting study objects for various disciplines, in sciences as well as in humanities. In particular, historical texts can provide rewarding starting points for natural product…
expired, 03/2021 - 10/2022
-
Development of small-molecules targeting CD93-signaling for the treatment of leukemia
The aim of the current InnoSuisse application is to optimize small molecules, which were identified in a drug library screen to block CD93-signaling in LSCs and showed a positive clinical response, in order to develop lead candidates with a novel chemical scaffold and own IP to treat leukemia.
expired, 01/2021 - 07/2022
-
Inflammasome inhibitors - Development of new inhibitors
Inhibition of the Inflammasome, a high-molecular complex, is a promising strategy to fight certain diseases with an (auto-)immune component, including gout, arthritis, Alzheimer's and Parkinson's.
expired, 06/2020 - 04/2022
-
Burn Wound Treatment
This project is evaluating inhibitors of matrix metalloproteinase 9 (MMP-9) for the treatment of burn wounds. A specific molecule that reduces the production and activity of MMP-9 in certain cells is being studied.
expired, 04/2020 - 09/2021
-
Silent pathway awakening to discover novel antibacterial compounds from actinomycetes
There is a tremendous need for novel antibiotics due to the global resistance crisis. Actinomycetes are the most important sources of secondary metabolites. They provide an enormous potential to discover novel antibiotics.
expired, 08/2019 - 01/2021
-
A topical anti-infective
ZHAWOC6027 ist a novel drug candidate against skin infections. In order to use it successfully, various properties need to be optimised.
expired, 05/2019 - 04/2021
-
Morbillivirus cell entry machinery: mechanisms, structures and antiviral drug discovery
expired, 04/2019 - 03/2025
-
Identification and characterization of active substances from actinomycetes from Switzerland against pathogenic bacteria
Actinomycetales is an order within the phylum Actinobacteria. Many actinomycetes produce antibiotics. A recently discovered new antibiotic, formicamycin from Streptomyces formicae, shows good activity against MRSA bacteria.
expired, 05/2018 - 12/2018
-
Novel Antimicrobials for Life-Threatening Infections
Deaths due to antimicrobial resistance are alarmingly high, novel therapies are urgently needed. Here, novel antifungal compounds are developed against life-threatening infections. The compounds will be developed to preclinical proof-of-concept in a collaborative medicinal chemistry program.
expired, 04/2018 - 09/2019
Selected publications
- Antiprotozoal Structure–Activity Relationships of Synthetic Leucinostatin Derivatives and Elucidation of their Mode of Action / M. Brand, L. Wang, S. Agnello, S. Gazzola, F. M. Gall, L. Raguž, M. Kaiser, R. S. Schmidt, A. Ritschl, J. Jelk, A. Hemphill, P. Mäser, P. Bütikofer, M. Adams, R. Riedl, Angew. Chem. Int. Ed. 2021, 60, 15613.
- Drug Design Inspired by Nature: Crystallographic Detection of an Auto‐Tailored Protease Inhibitor Template / F. M. Gall, D. Hohl, D. Frasson, T. Wermelinger, P. R. E. Mittl, M. Sievers, R. Riedl, Angew. Chem. Int. Ed. 2019, 58, 4051.
- A Structural View on Medicinal Chemistry Strategies against Drug Resistance / S. Agnello, M. Brand, M. F. Chellat, S. Gazzola, R. Riedl, Angew. Chem. Int. Ed. 2019, 58, 3300.
- Pseudouridimycin: The First Nucleoside Analogue That Selectively Inhibits Bacterial RNA Polymerase / M. F. Chellat, R. Riedl, Angew. Chem. Int. Ed. 2017, 56, 13184.
- Targeting Antibiotic Resistance / Chellat, Mathieu; Raguž, Luka; Riedl, Rainer - Angew. Chem. Int. Ed. 2016, 55, 6600-6626; Angew.Chem. 2016, 128, 6710–6738.
- Molecular recognition of the catalytic zinc (II) ion in MMP-13: Structure-based evolution of an allosteric inhibitor to dual binding mode inhibitors with improved lipophilic ligand efficiencies / Fischer, Thomas; Riedl, Rainer - invited article for the Special Issue "Enzyme-Inhibitor Interaction as Examples of Molecular Recognition" Int. J. Mol. Sci. 2016, 17, 314. Front cover story 3/2016.
- Merging Allosteric and Active Site Binding Motifs: De novo Generation of Target Selectivity and Potency via Natural-Product-Derived Fragments / Lanz, Jan; Riedl, Rainer - ChemMedChem. 2015, 10, 451–454. Front cover story 3/2015.
Publications
-
Scherer, Melanie; Djabeur, Nadia; Siering, Oliver; Jeckelmann, Jean-Marc; Wyss, Marianne; Cresci, Marina; Di Palma Subran, Morgane; Riedl, Rainer; Chames, Patrick; Pfaller, Christian K.; Sawatsky, Bevan; Fotiadis, Dimitrios; Plattet, Philippe,
2026.
Nature Communications.
17(1), pp. 103.
Available from: https://doi.org/10.1038/s41467-025-67600-z
-
Nasi, Sonia; Ehirchiou, Driss; Blatter, Cindy; Chobaz, Veronique; Germann, Stefan; Brandenberger, Alexandra; Bommeli, Elias; Uchikawa, Emiko; Cirino, Giuseppe; Riedl, Rainer; So, Alexander; Busso, Nathalie,
2025.
Pharmacological Research.
219(107869).
Available from: https://doi.org/10.1016/j.phrs.2025.107869
-
Ehirchiou, Driss; Nasi, Sonia; Chobaz, Véronique; Riedl, Rainer; Germann, Stefan; Brandenberger, Alexandra; Bommeli, Elias; Blatter, Cindy; Cirino, Giuseppe; So, Alexander; Busso, Nathalie,
2025.
In:
Annual European Congress of Rheumatology EULAR, Barcelona, Spain, 11-14 June 2025.
Elsevier.
pp. 112-113.
Available from: https://doi.org/10.1016/j.ard.2025.05.149
Awards & News
ZHAW News-Release 2018: Bachelor thesis on structural optimization of a matrix metalloproteinase-13 inhibitor is awarded Dr. Max Lüthi Prize of the SCS.
ZHAW News-Release 2016: Bachelor thesis on rational drug design and synthesis of cyclic peptides is awarded Dr. Max Lüthi Prize of the SCS.
Poster(PDF 891,1 KB) 2013: SCG-FH Award Category «Molecules for the Life Sciences» Tackling antibiotic resistance: A joint project of the center for organic and medicinal chemistry ZHAW and the Bioversys AG.
ZHAW press release(PDF 74,7 KB) (German) 2013: SCS awarded the Dr. Max Lüthi award for bachelor thesis on the rational design and synthesis of novel protease inhibitors.
ZHAW press release(PDF 46,8 KB) (German) 2012: MSc thesis on the structure based design and synthesis of novel protease inhibitors received Bodenseeinnovation award.
ZHAW press release (PDF 46,4 KB)(German) 2011: Bachelor thesis on development of active substances to combat antibiotic resistance is awarded Dr. Max Lüthi award of the SCS.